2021 Project: Feedback and Oscillations: Engineering control systems for diabetes therapy
Faculty Director: Dr. Ben Cosgrove, Assistant Professor
Department: Meinig School of Biomedical Engineering, Cornell University
 Dr. Ben Cosgrove is an Assistant Professor in the Meinig School of Biomedical Engineering at Cornell University in Ithaca, NY, where he directs the Laboratory of Regenerative Systems Biology. Dr. Cosgrove earned a Bachelor’s in Biomedical Engineering at the University of Minnesota, a Ph.D. in Bioengineering at the Massachusetts Institute of Technology under the supervision of Dr. Douglas Lauffenburger and Dr. Linda Griffith, and completed a postdoctoral fellowship at the Stanford University School of Medicine with Dr. Helen Blau. To learn more about Dr. Cosgrove, visit his faculty profile.
Dr. Ben Cosgrove is an Assistant Professor in the Meinig School of Biomedical Engineering at Cornell University in Ithaca, NY, where he directs the Laboratory of Regenerative Systems Biology. Dr. Cosgrove earned a Bachelor’s in Biomedical Engineering at the University of Minnesota, a Ph.D. in Bioengineering at the Massachusetts Institute of Technology under the supervision of Dr. Douglas Lauffenburger and Dr. Linda Griffith, and completed a postdoctoral fellowship at the Stanford University School of Medicine with Dr. Helen Blau. To learn more about Dr. Cosgrove, visit his faculty profile.
Project Background
Diabetes is a disorder characterized by hyperglycemia or elevated blood glucose it is the most common endocrine disorder. Thirty million Americans have diabetes, yet many are not aware of it. Diabetics must adjust their intake of carbohydrates and their dose of insulin, in order to maintain blood glucose levels that are near normal. Diabetes has potential long-term complications that can affect the kidneys, eyes, heart, blood vessels, and nerves. These complications appear to be related to the degree of control that the diabetic can maintain over their glucose level. There are two types of diabetes, Type 1 and Type 2. Type 1 diabetics do not produce insulin due to an autoimmune response which attacks insulin-producing pancreatic cells. People with Type 2 diabetes can still make insulin however, their cells have some degree of insulin resistance i.e. the muscle, fat, and liver cells do not respond to insulin signals to remove glucose. This results in a build-up of glucose. Initial therapy for Type 2 diabetics with mild disease is lifestyle modification: a healthy diet with exercise to help lose weight. If this fails, medication is recommended that increases the number of insulin receptors present on cells, so they become more sensitive to insulin.
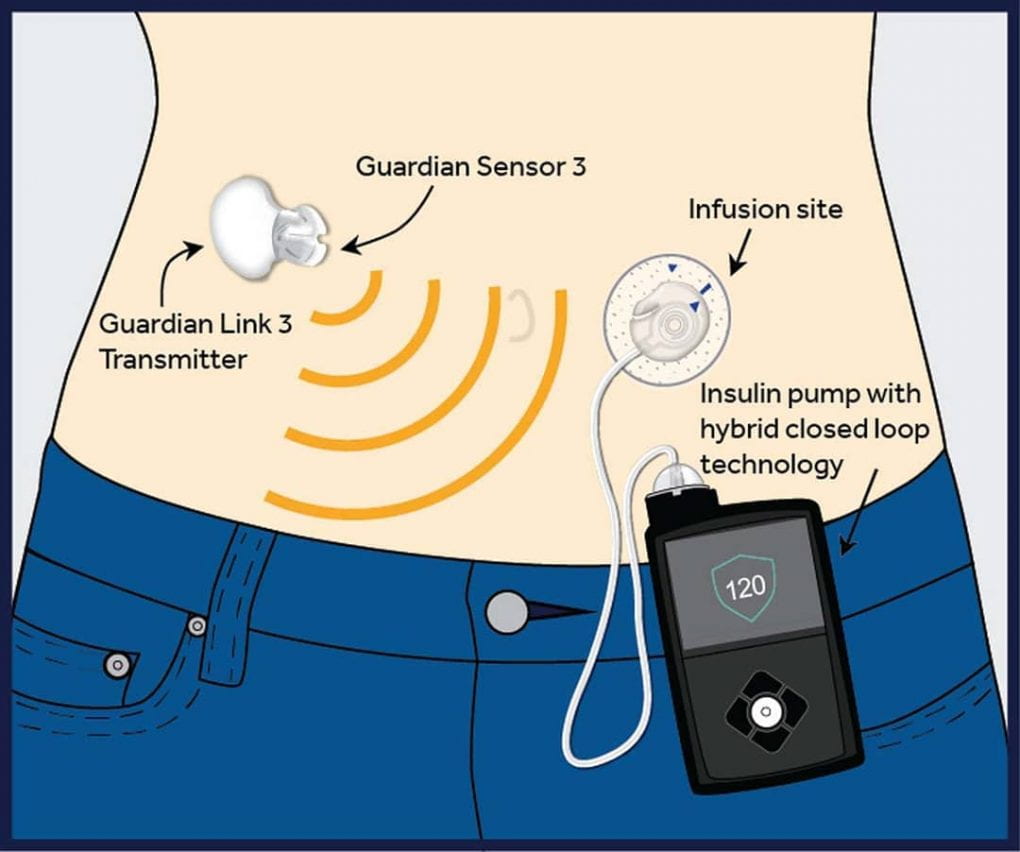
The only effective treatment in Type 1 diabetes is administering insulin as these patients no longer produce it. There are many different types of insulin and different regimens, but many patients will use long-acting insulin at night supplemented by short-acting insulin before mealtimes. Newer treatment regimens include the use of an insulin pump where blood glucose levels are entered into a machine which then uses an algorithm to pump insulin into the body. In 2016 the FDA approved the first insulin pumps that mimic the way the human pancreas works by delivering small doses of short-acting insulin continuously. The device also can deliver variable amounts of insulin when a meal is eaten. This has been a huge advancement in diabetic care. Other novel treatments being investigated are a combination of tissue engineering and biomaterial approaches to produce functioning pancreatic cells e.g. using stem cells that can act as a bioartificial pancreas. A bioartificial pancreas is a device that can be implanted into the patient and take over the function of insulin/glucose regulation, it contains synthetic materials and functional islet cells encapsulated within a semipermeable membrane to protect them from immunological rejection. The encapsulated islets can act as immune barriers while permitting mass transfer of nutrients, glucose, and insulin to and from the islets. Each of these therapies requires careful balancing of insulin and nutrient levels to maintain glucose balance throughout the day. Therefore, they provide a challenging dynamic system of feedback control mechanisms that necessitate engineering control measures to stabilize treatment strategies and design long-term insulin administration strategies to achieve desired benefits to the patients.
In the week-long CATALYST project, students will learn about various bioengineering approaches to diabetes treatment. We will learn about bioengineered therapies, both in the developmental stages and clinical use and also design new and better strategies. Students will learn the principles of dynamic control systems and the mathematical and computational skills (using MATLAB) to implement them to understand human physiology and design better therapies. Students will use computational approaches to model new diabetes treatment strategies and be enlisted in a design competition to generate the next generation of insulin delivery technologies.
Previous Projects
Information about projects from previous years can be found below: